The ProjectFor this project, we had to make a board game that incorporated chemical reactions. Instead of the usual group of four, we decided to make a joint game with another group to form a group of eight. After some brainstorming, we came up with a Toy Story theme and called our game ¨Andy's Coming." The game is based on the original Toy Story movie where the toys get stuck at Sid's house (the purple, bottom board); the object of the game is to get all the toys back to Andy's house (the blue, top board) and safely into the toy chest, without getting caught, before Andy comes home. The game has cards that tell players how many spaces to move or direct them where to go. If a player lands on a star, the player must pick up a star card that features a storyline and required chemical reaction. We included seven reaction cards: two single replacement reactions+lighting of an LED, three double displacement reactions, one production of gas, and one combustion reaction. The chemicals used and the descriptions of all the reactions can be found below.
|
Content
Game set up/rules/safety:
Reactions:
Single Displacement Reaction
The anion of one polyatomic ion moves to the other cation: A+BX ->AX+B
In our project one of our single replacement reactions was the lighting of the LED. Two moles of Aluminum reacts with three moles of Copper (II) Chloride to produce three moles of Copper and two moles of Aluminum (III) Chloride.
2 Al(s) + 3 CuCl2 (aq) --> 3 Cu (s) +2 AlCl3
In our project one of our single replacement reactions was the lighting of the LED. Two moles of Aluminum reacts with three moles of Copper (II) Chloride to produce three moles of Copper and two moles of Aluminum (III) Chloride.
2 Al(s) + 3 CuCl2 (aq) --> 3 Cu (s) +2 AlCl3
Double Displacement Reaction
The anions of both polyatomic ions switch cations: AX + BY ->AY+BX
In our project one double displacement reaction was creating the color brown. Three moles of Potassium Chromate reacts with 2 moles of Iron (III) Chloride to produce one mole of Iron (III) Chromate and six moles of Potassium Chloride.
3 K2CrO4 (aq) + 2 FeCl3 (aq) --> Fe2(CrO4)3 (s) + 6 KCl (aq)
In our project one double displacement reaction was creating the color brown. Three moles of Potassium Chromate reacts with 2 moles of Iron (III) Chloride to produce one mole of Iron (III) Chromate and six moles of Potassium Chloride.
3 K2CrO4 (aq) + 2 FeCl3 (aq) --> Fe2(CrO4)3 (s) + 6 KCl (aq)
Combustion Reaction
A hydrocarbon and oxygen combine to form carbon dioxide and water: CH+O2 -->CO2 +H2O
In our project the combustion reaction was to create a flame. For every mole of ethanol you get three moles of water.
C2H5OH(l) + 3O2(g) --> 2CO2(g) + 3H2O(l)
In our project the combustion reaction was to create a flame. For every mole of ethanol you get three moles of water.
C2H5OH(l) + 3O2(g) --> 2CO2(g) + 3H2O(l)
Production of a Gas
A combination of a carbonate and a hydrochloric acid.
In our project our production of a gas was blowing up a balloon using a combination of a carbonate and a hydrochloric acid.
CaCO3(s) + 2HCI(aq) --> CaCI2(aq) + CO2(g) + H2O(l)
In our project our production of a gas was blowing up a balloon using a combination of a carbonate and a hydrochloric acid.
CaCO3(s) + 2HCI(aq) --> CaCI2(aq) + CO2(g) + H2O(l)
cation
A positive, often metal ion.
anion
A negative, often negative ion.
Reactant
A substance that takes part in and undergoes change during a reaction.
Product
A substance that is formed as the result of a chemical reaction.
synthesis
Adding two ions: A+B -> AB
Decomposition
Taking apart two ions: AB -> A+B
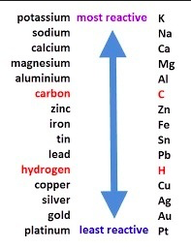
Reactivity series
Reflection
I enjoyed doing this project because it allowed us to be creative while also putting what we're learning to real-world use. I also liked that our group combined with another group, which allowed for even more creative ideas. In my original group of four, we were all fairly chatty and easily distracted, and it was hard for us to get back on task. Our group had many different ideas, and we didn't have great communication skills, so there were occasional frustrations when things didn't go as expected. Some group members were also absent during some parts of the building process, so we were often on different pages to one another. We also didn't manage our time well, and spent too much of our time decorating the board. We ended up doing the bulk of the work in the last few class sessions, and I ended up making the cards and game pieces on my own at home. The group we eventually collaborated with was nice and helpful. The combined group was a lot of fun, but it presented a challenge at presentation time. Because there were so many of us, our planned parts were covered unexpectedly by other group members; in the typical four-person group, every member would have a featured speaking role during the presentation. As for positives in this project, we were all relieved and happy with our work when it all came together. I also learned a lot about the different chemical reactions, and it was fun to actively make them occur on our game board. It was much like the Rube Goldberg project that we did our freshman year of STEM, which I also enjoyed. Our theme was clever and fun, and I liked how we brought some of the storyline into the game. Overall, this project has taught me many life skills, including staying focused, being more patient with others and staying positive rather than getting frustrated. I am excited to see what's in store for the rest of the year.